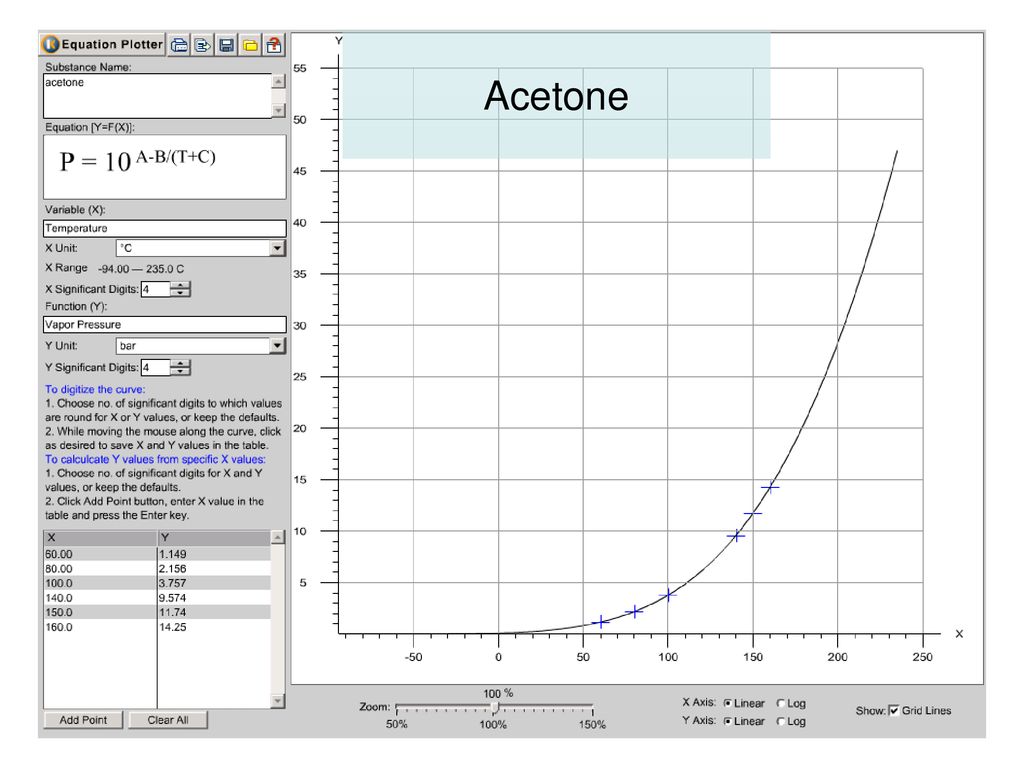
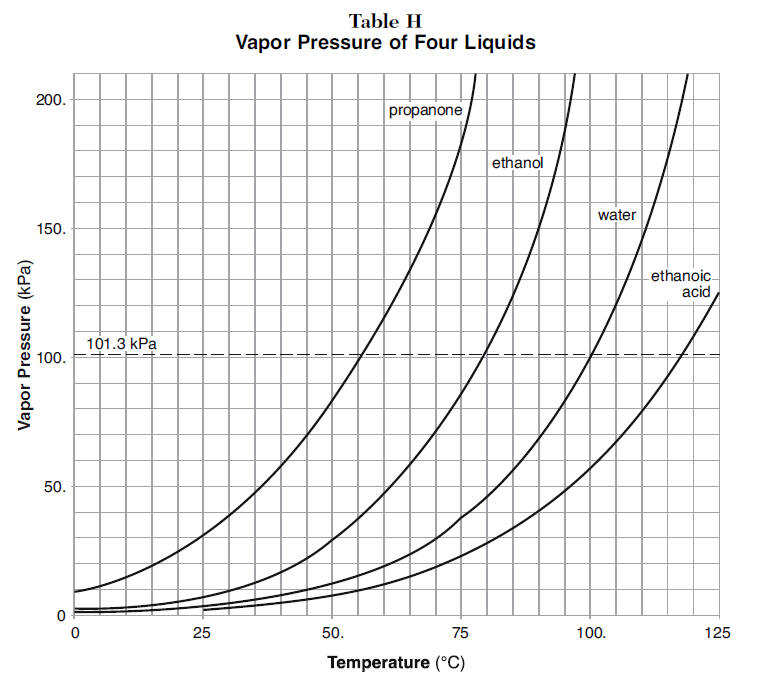
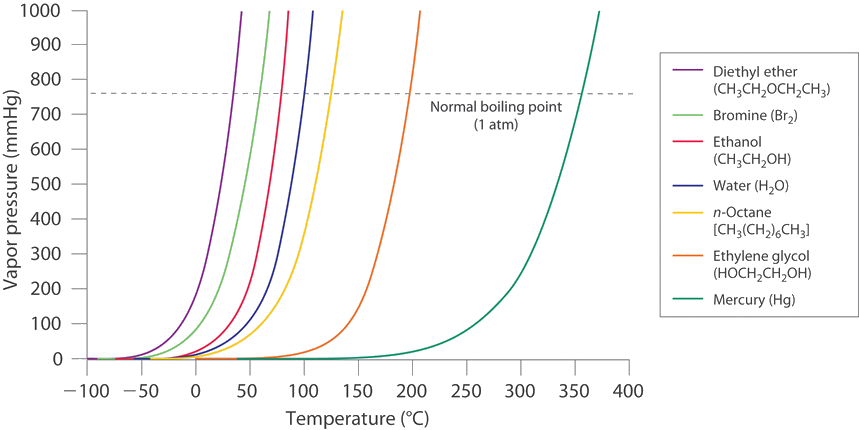
The vapor pressure of acetone at `20^(@)C` is 185 torr. When `1.2 g` of a non-volatile solute - YouTube

The saturation vapor pressure of the benzene/acetone mixture (C 6 H 6 +... | Download Scientific Diagram
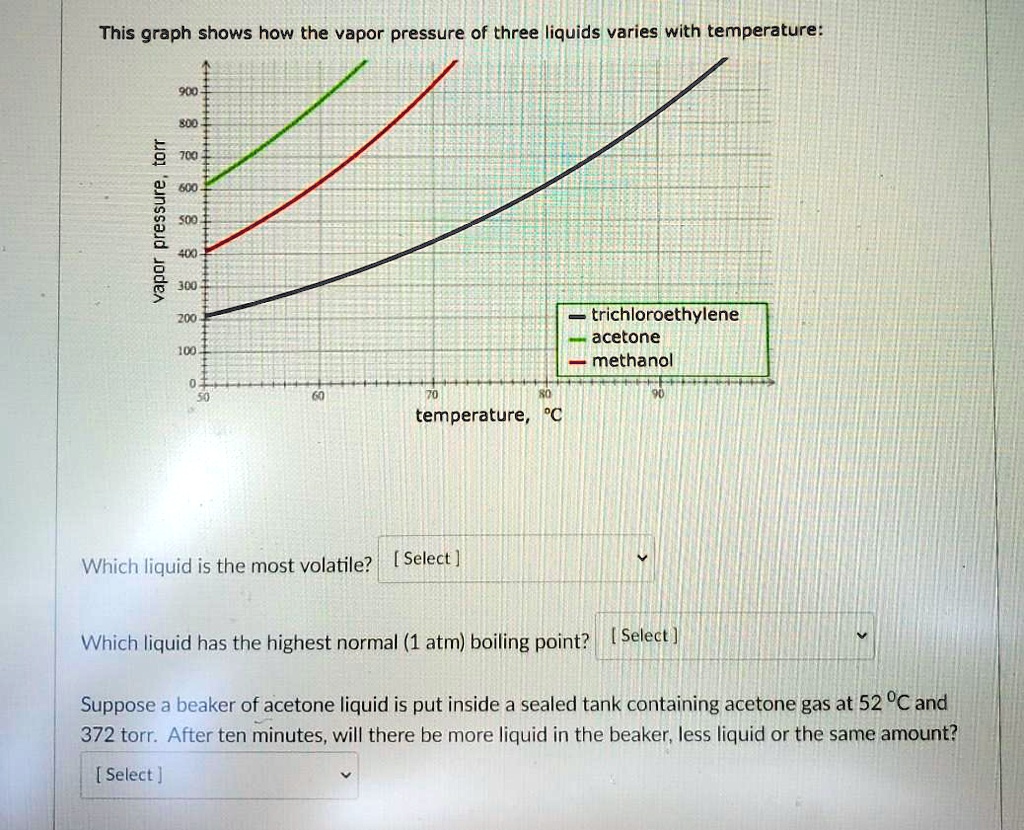
SOLVED: This graph shows how the vapor pressure of three liquids varies with temperature: 27.00 60.00 140.00 8.00 700.00 trichloroethylene acetone methanol 10.00 temperature, "C Which liquid is the most volatile? Select

Use the following vapor pressure diagram to estimate the partial pressure of acetone. | Homework.Study.com
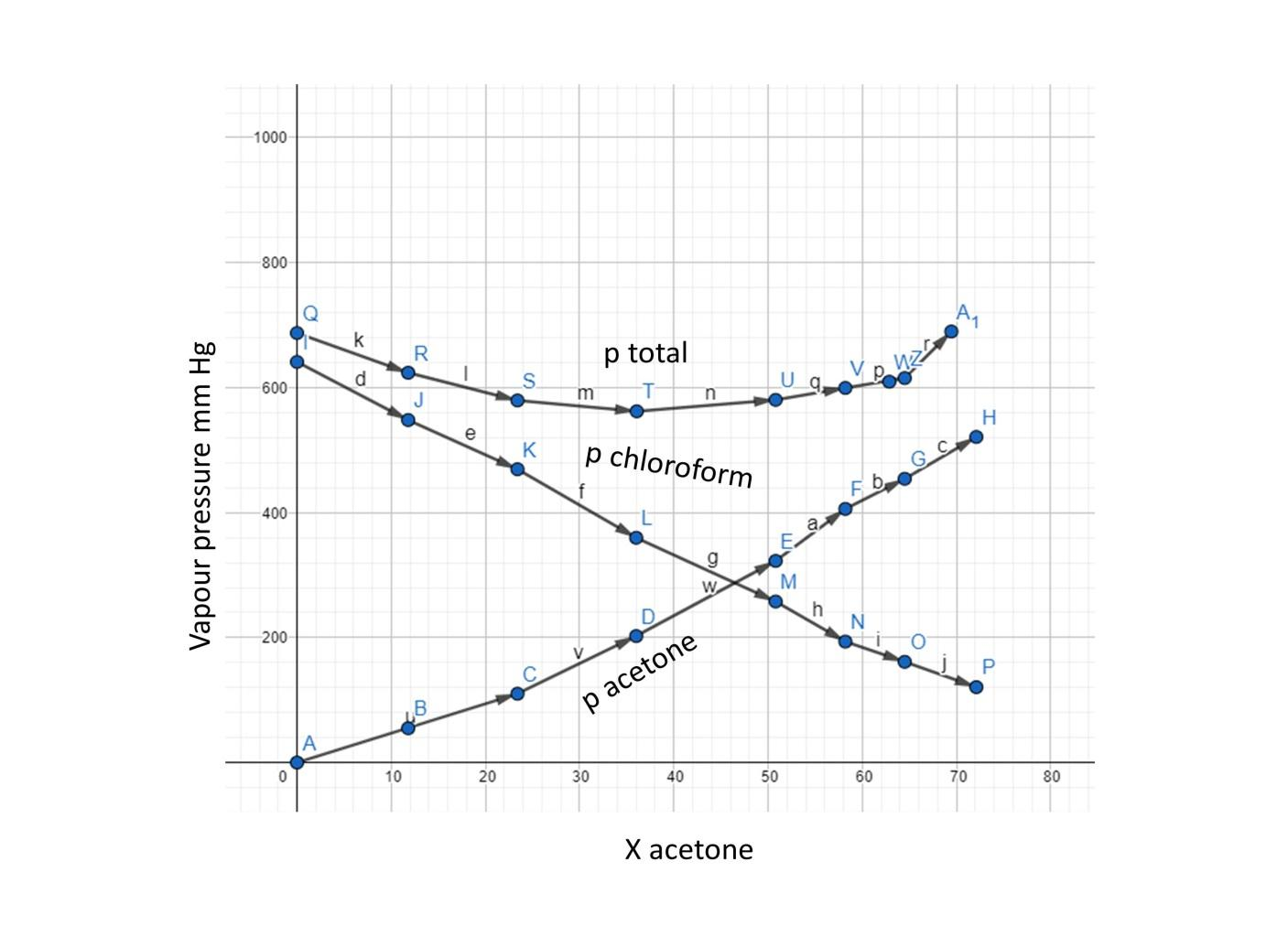
Vapour pressure of pure acetone and chloroform at $328\\,K$ are $741.8mmHg$ and $632.8mmHg$ respectively. Assuming that they form an ideal solution over the entire range of composition. Plot ${P_{total}},{P_{chloroform}}$ and ${P_{acetone}}$ as
Equilibrium phase diagram of acetone/ethanol mixture under the pressure... | Download Scientific Diagram