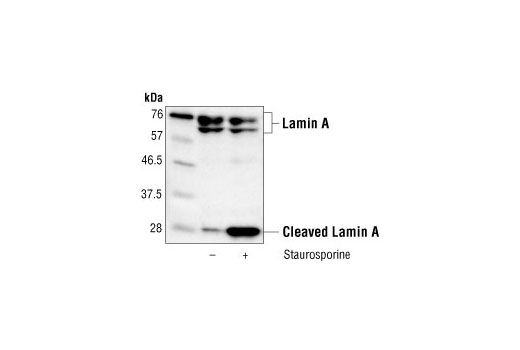
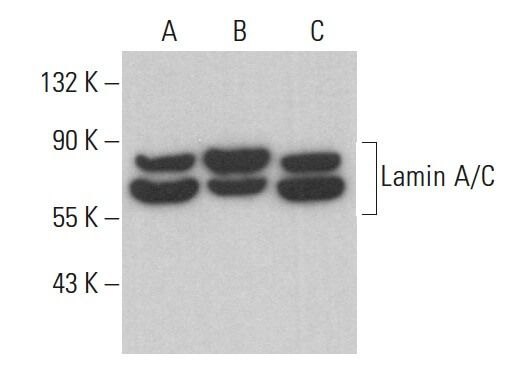
Role of nuclear Lamin A/C in cardiomyocyte functions - Carmosino - 2014 - Biology of the Cell - Wiley Online Library

Matrix Elasticity Regulates Lamin-A,C Phosphorylation and Turnover with Feedback to Actomyosin: Current Biology
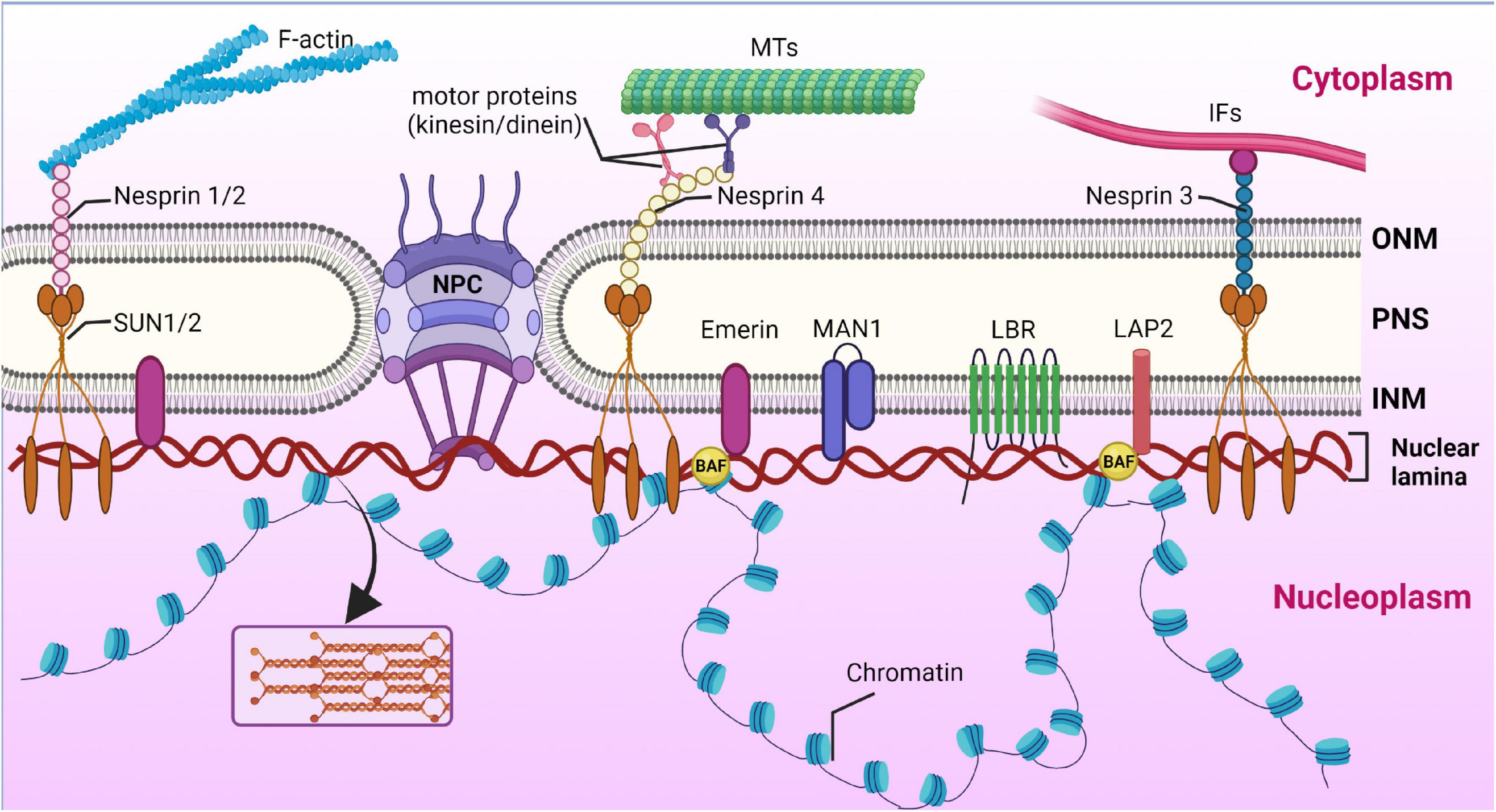
Frontiers | Diversity of Nuclear Lamin A/C Action as a Key to Tissue-Specific Regulation of Cellular Identity in Health and Disease
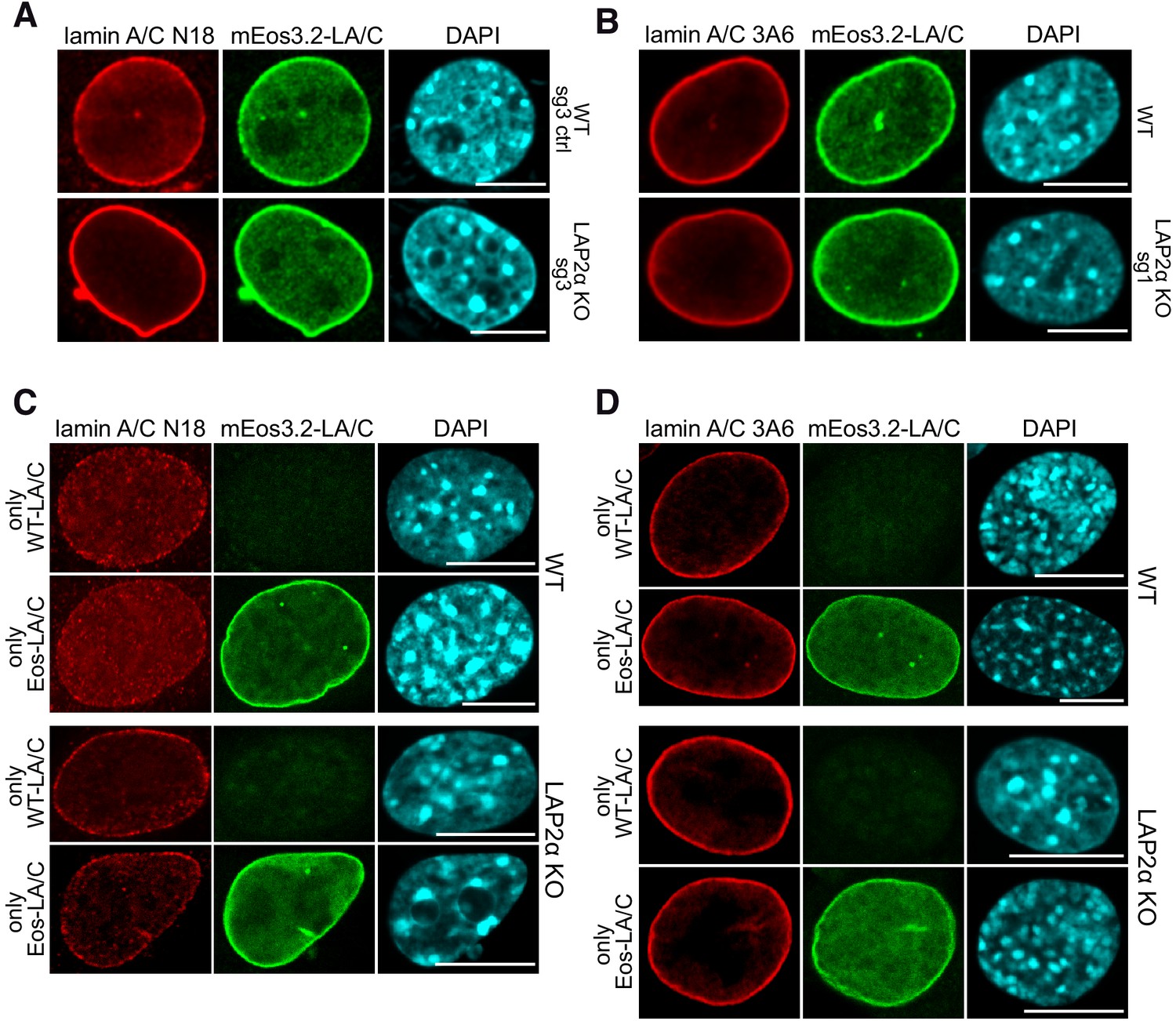
Figures and data in LAP2alpha maintains a mobile and low assembly state of A-type lamins in the nuclear interior | eLife
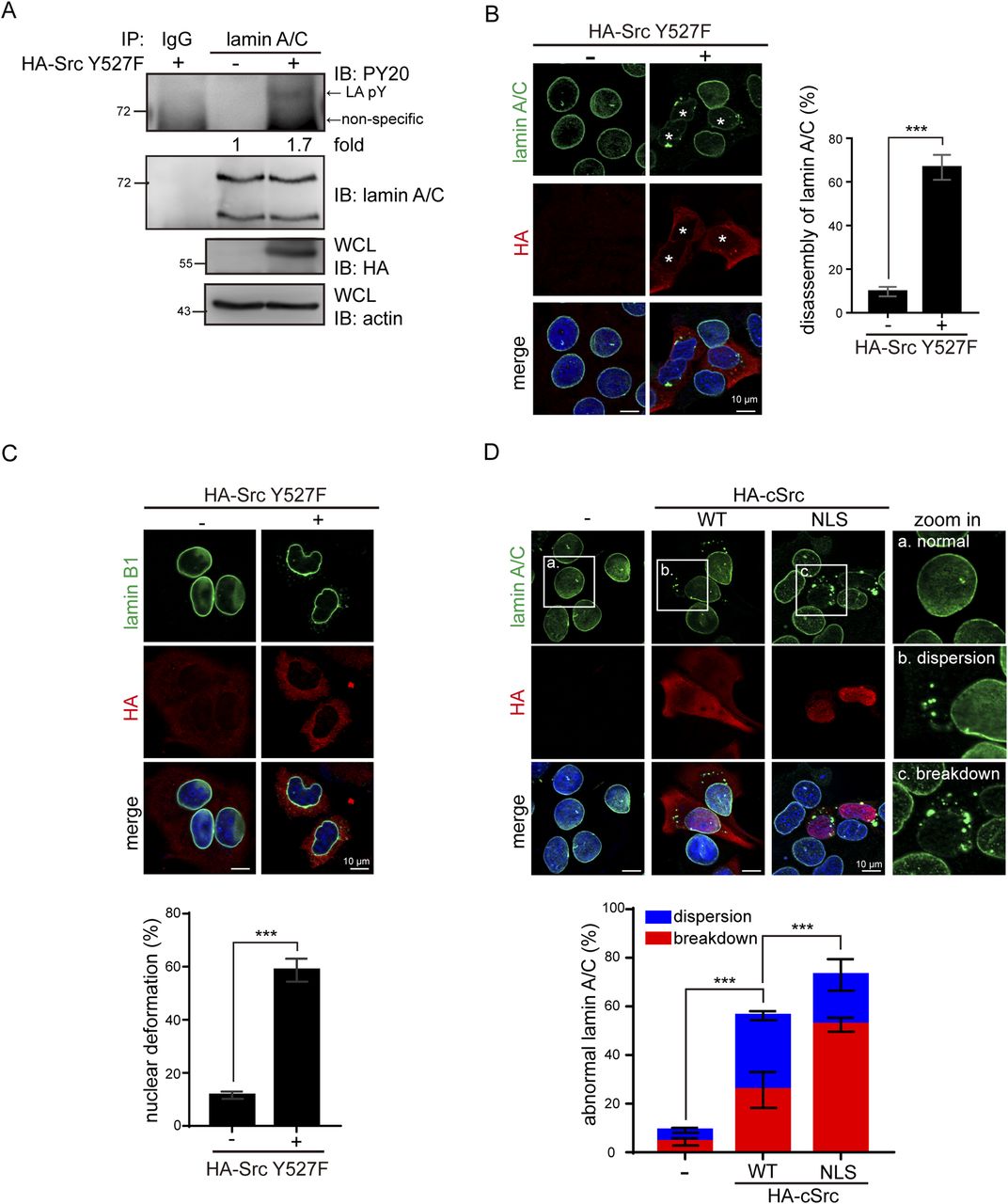
Tyrosine phosphorylation of lamin A by Src promotes disassembly of nuclear lamina in interphase | Life Science Alliance


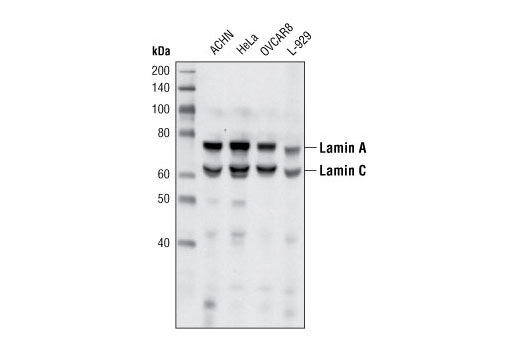

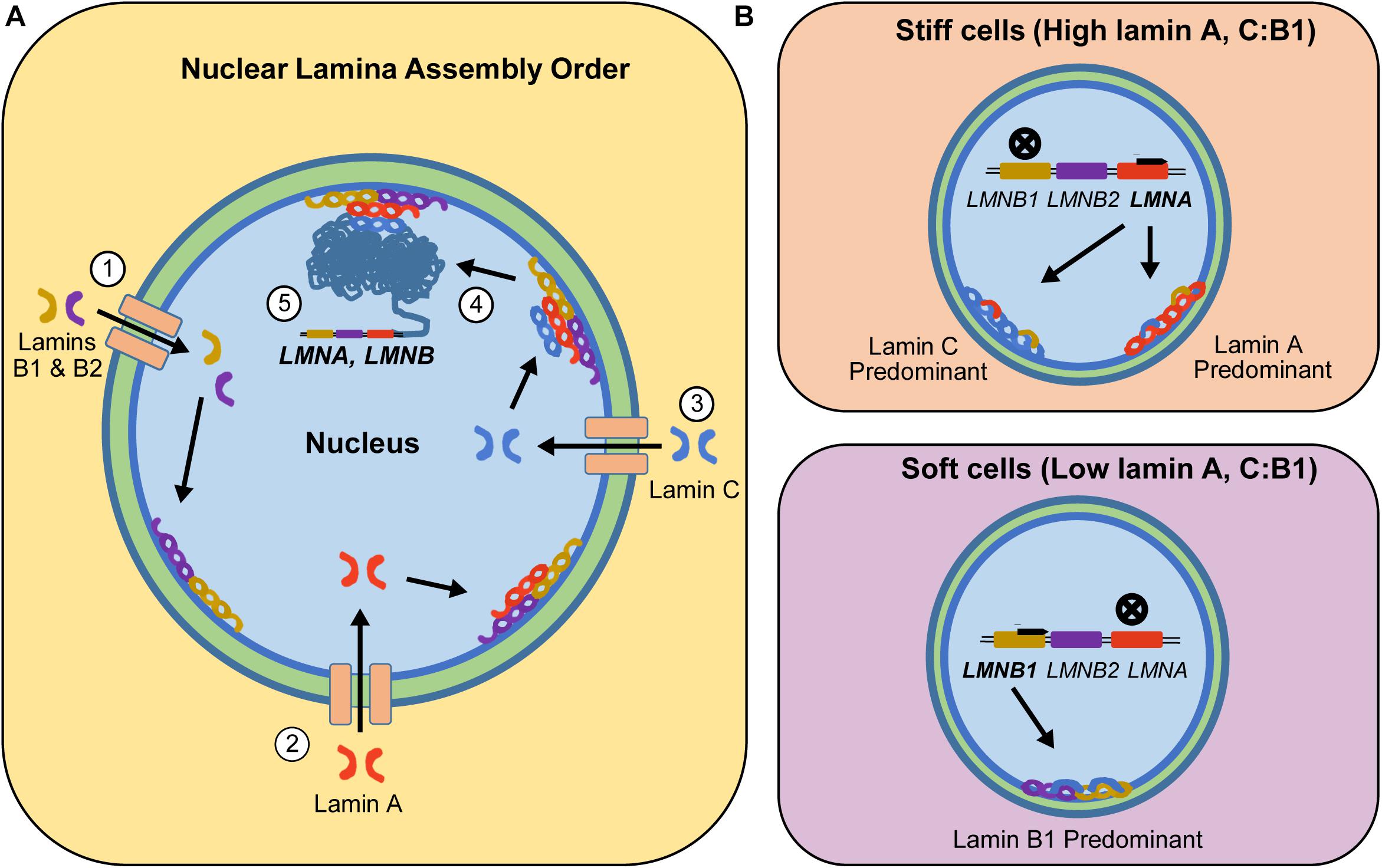
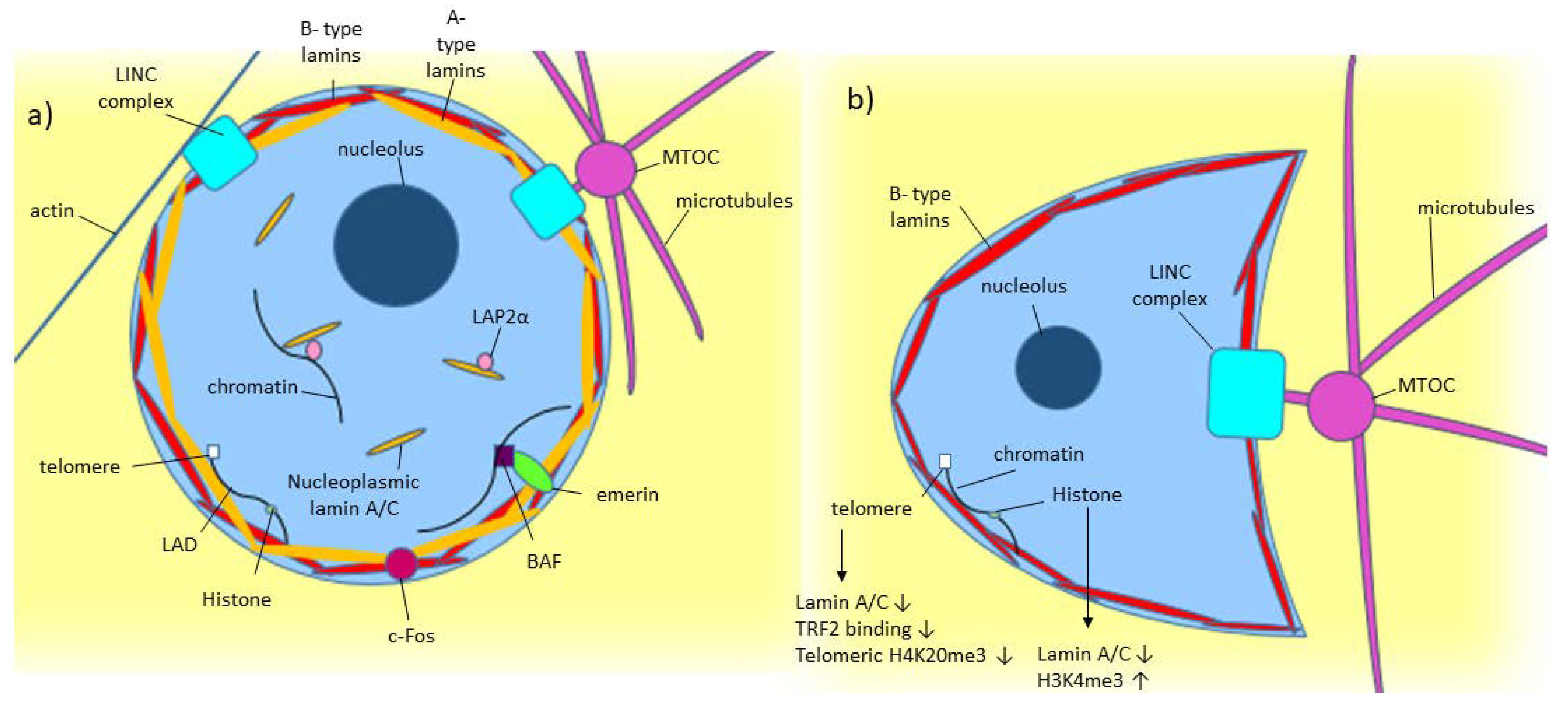
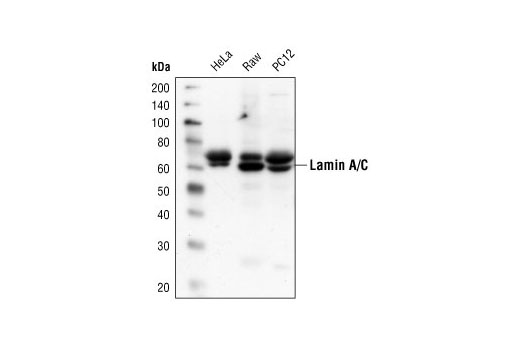
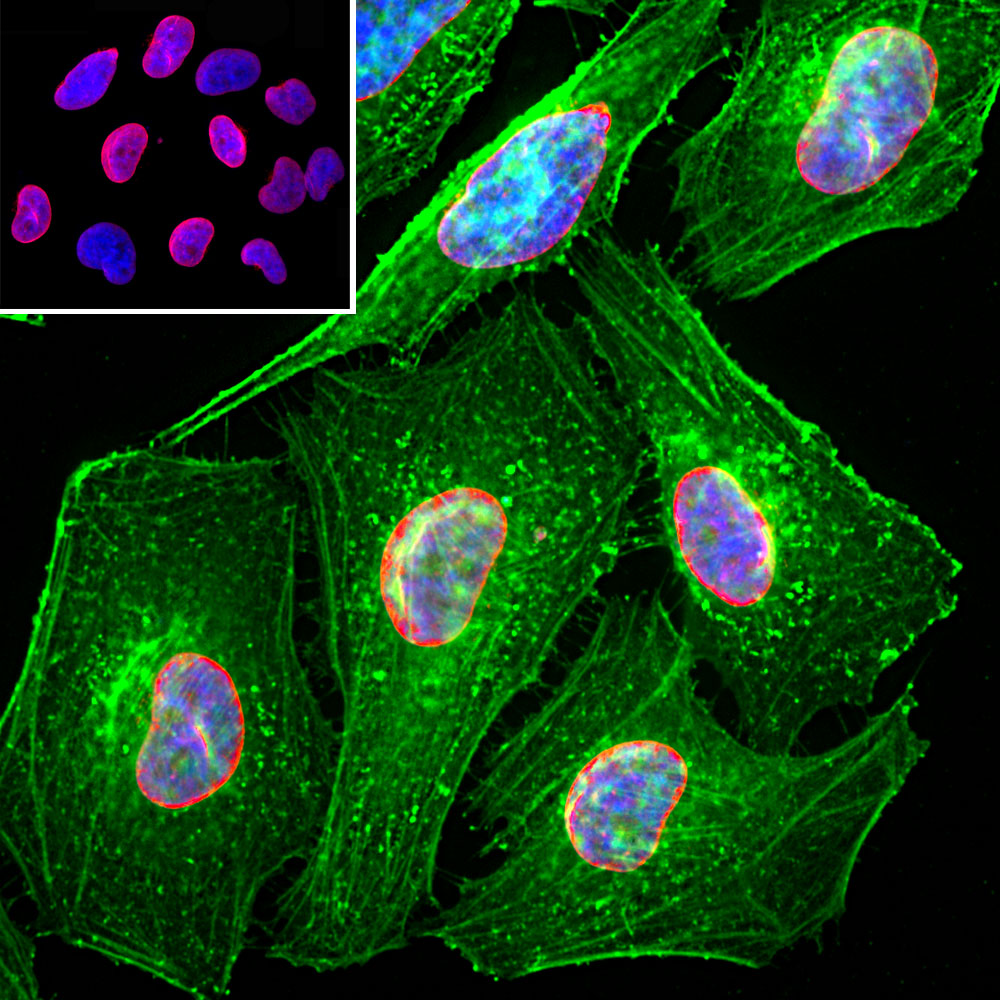


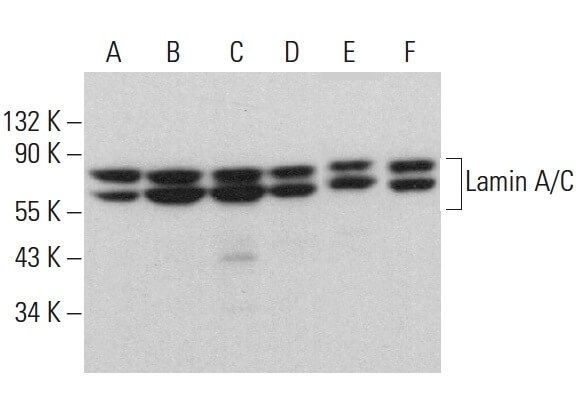
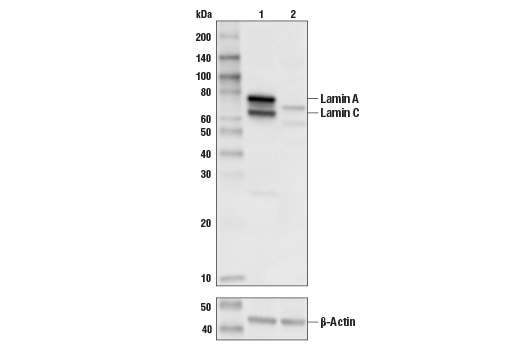
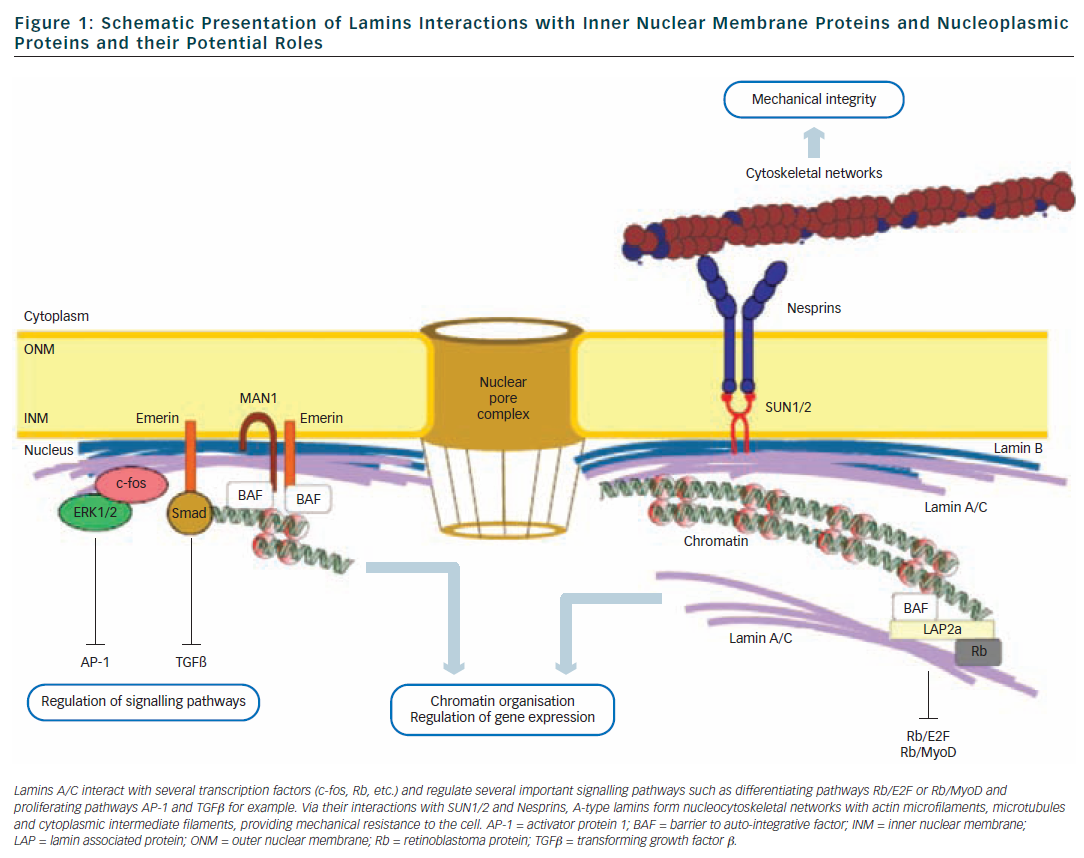
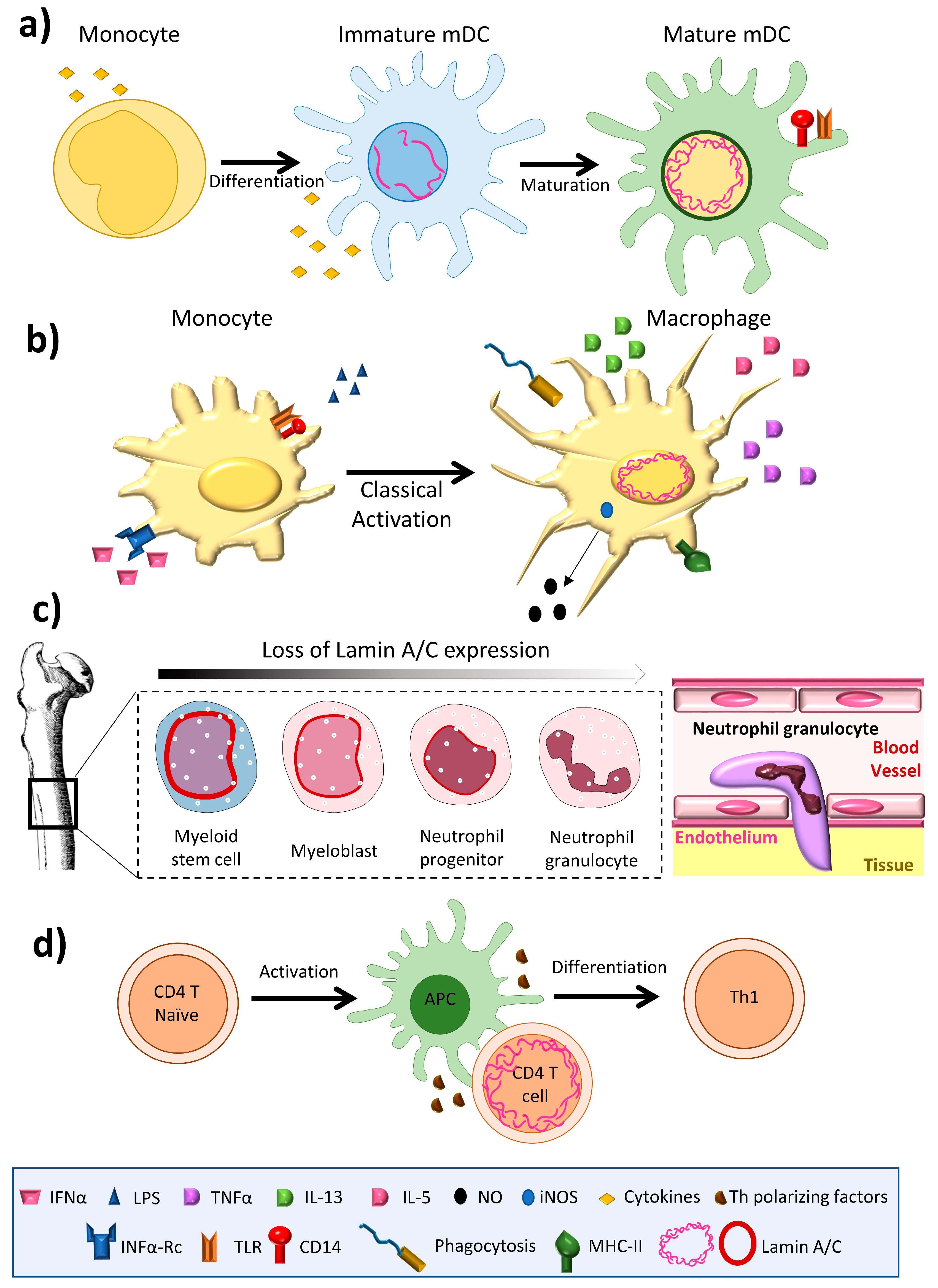


![Anti-Lamin A C [LASS2D9] | Monoclonal Antibodies - Ximbio Anti-Lamin A C [LASS2D9] | Monoclonal Antibodies - Ximbio](https://res.cloudinary.com/ximbio/image/upload/c_fit,fl_lossy,q_auto/fa521c9a-0d52-4e79-8d5b-1a1988e6e3b6.jpg)