Should be a Third Dose of BNT162b2 mRNA COVID-19-Vaccine Administered in Patients with Myelofibrosis Under Ruxolitinib? - ScienceDirect
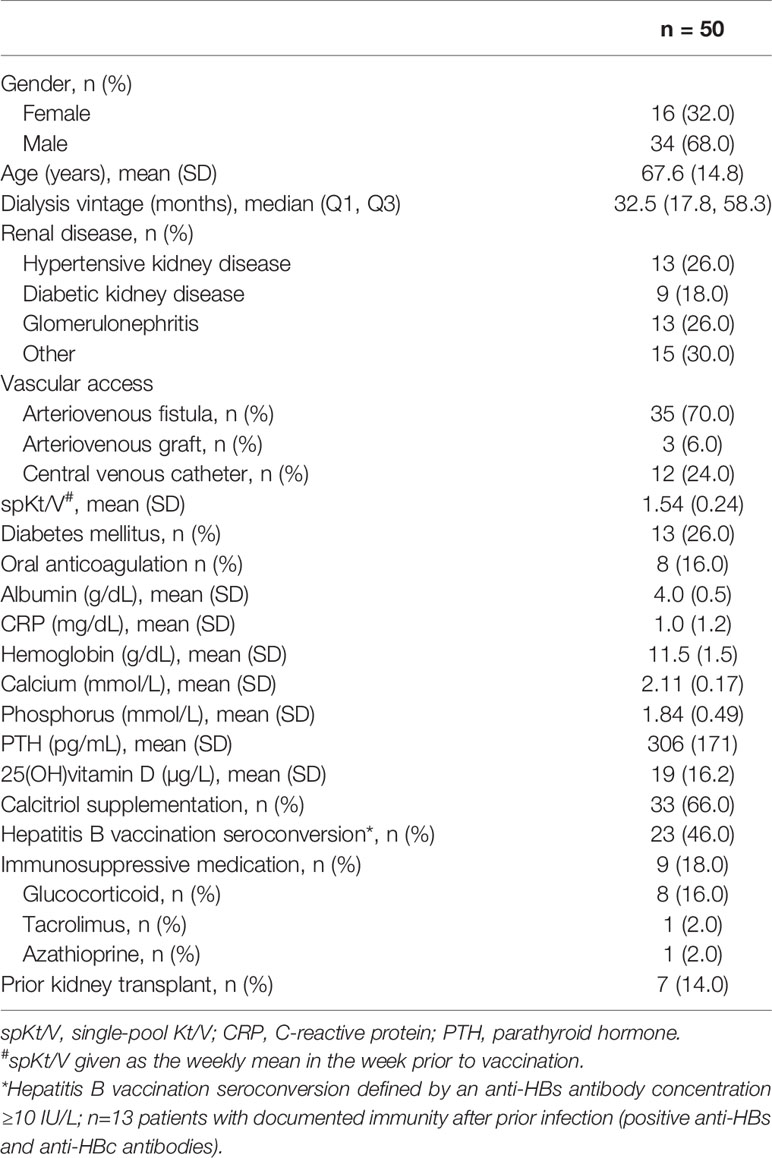
Frontiers | The Safety and Immunogenicity of the mRNA-BNT162b2 SARS-CoV-2 Vaccine in Hemodialysis Patients
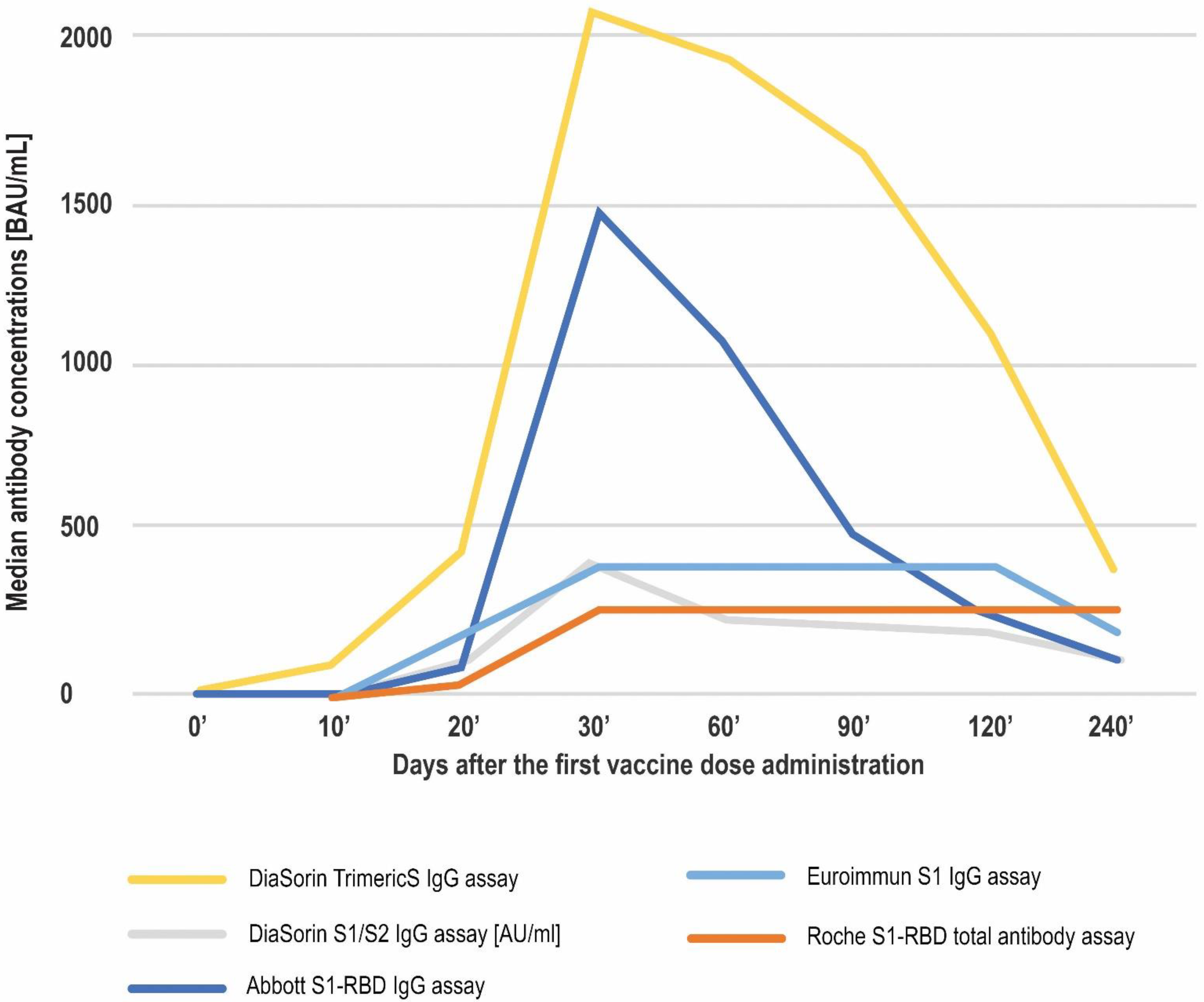
Diagnostics | Free Full-Text | Head-to-Head Comparison of 5 Anti-SARS-CoV-2 Assays Performance in One Hundred COVID-19 Vaccinees, over an 8-Month Course
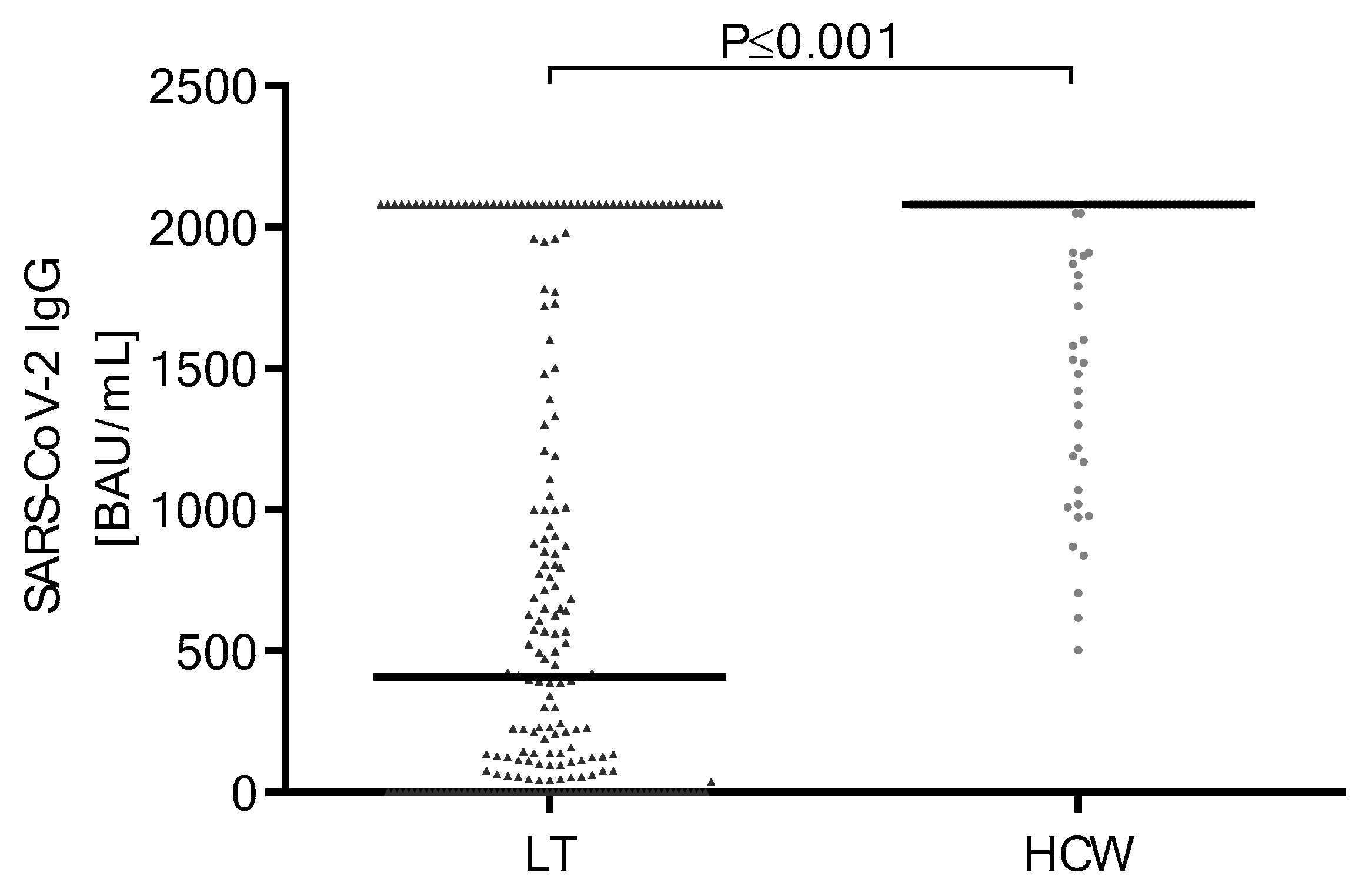
Vaccines | Free Full-Text | Durability of Immune Response after Application of a Third Dose of SARS-CoV-2 Vaccination in Liver Transplant Recipients

Estimating immunity with mathematical models for SARS-CoV-2 after COVID-19 vaccination | npj Vaccines

Evaluation of the analytical performance of three chemiluminescence serological assays for detecting anti-SARS-CoV-2 antibodies | Clinical and Experimental Medicine